Newsroom (42)
Publications
08/02/2022
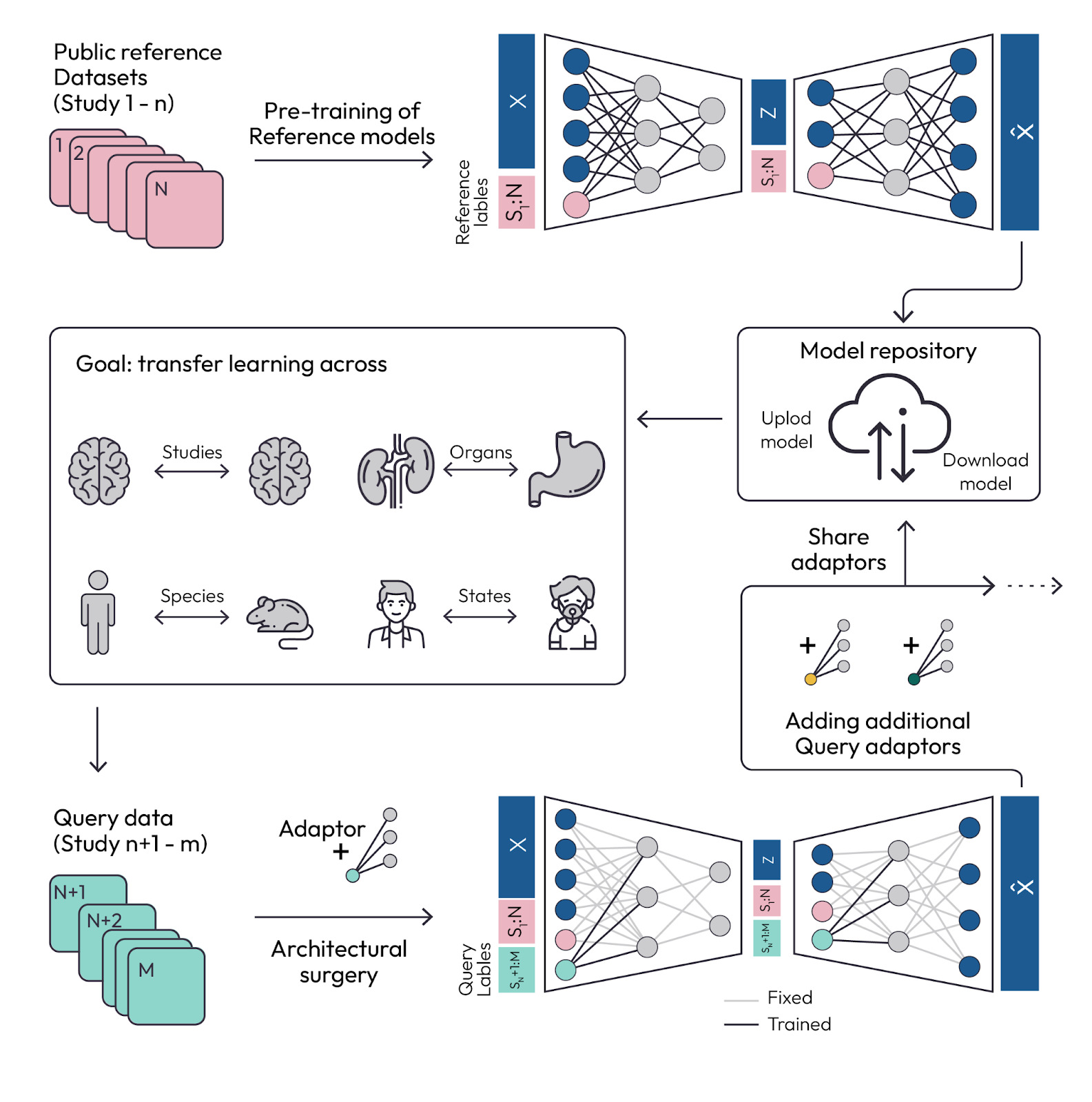
Quantifying the Reproducibility of Cell-Perturbation Experiments
Publications
07/26/2022
Neural Design for Genetic Perturbation Experiments
Publications
06/18/2022
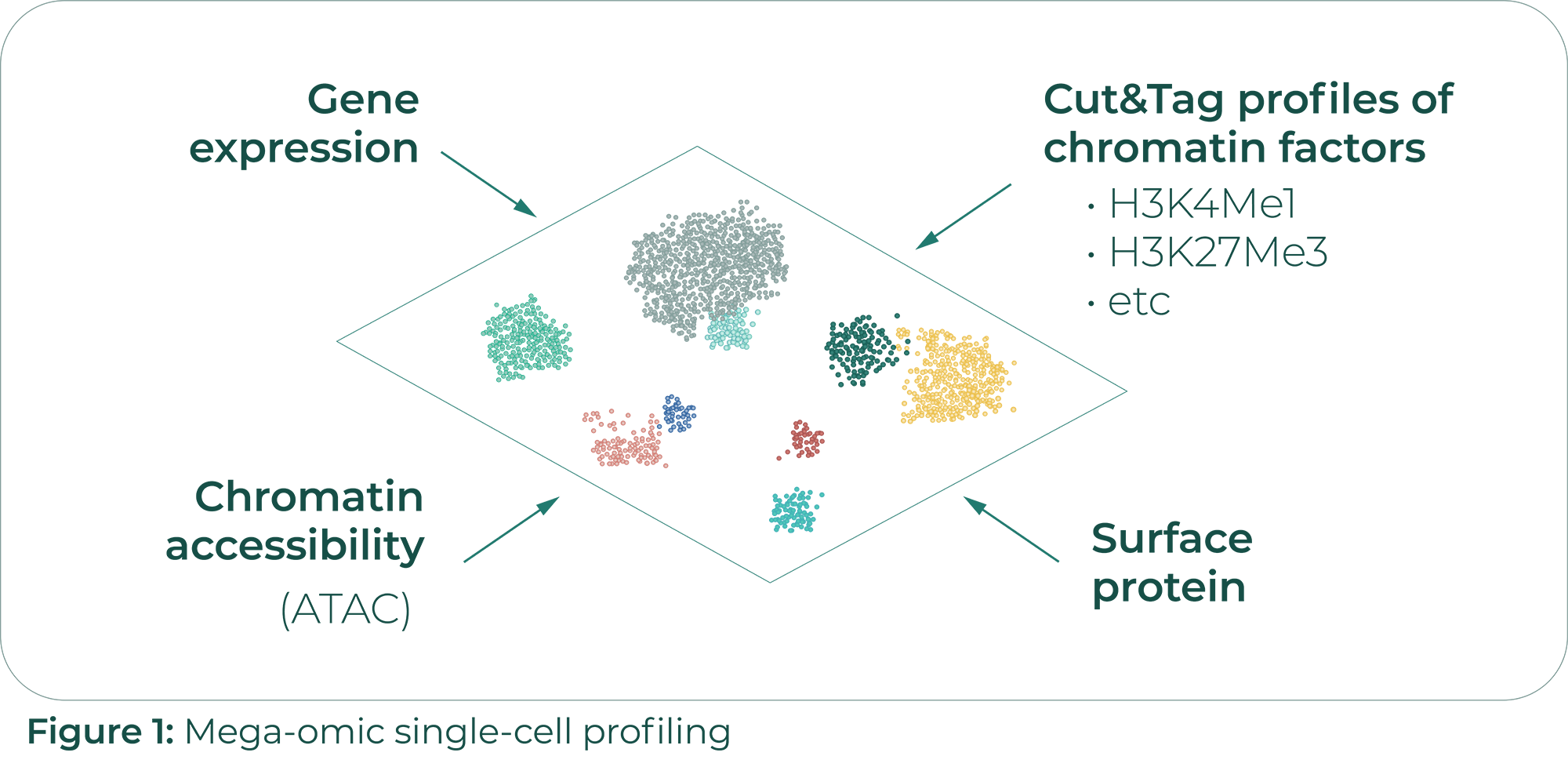
Massively parallel single-cell chromatin landscapes of human immune cell development and intratumoral T cell exhaustion
Publications
05/14/2022
Systemmatch: Optimizing Preclinical Drug Models To Human Clinical Outcomes Via Generative Latent-space Matching
Blog
02/27/2022
Advancements in Gene Regulatory Networks and Perturbation Predictions: Immunai’s Third Symposium
News
10/27/2021
Koch leads mammoth $215M infusion into AI startup boasting ‘largest database’ of immune cells
Press Releases
10/27/2021
Immunai Raises $215 Million to Accelerate Development of Its Immune-First Drug Actuary Platform
Blog
10/26/2021